What Is Data Provenance? Definition, Lineage, And Examples


What Is Data Provenance? Definition, Lineage, And Examples
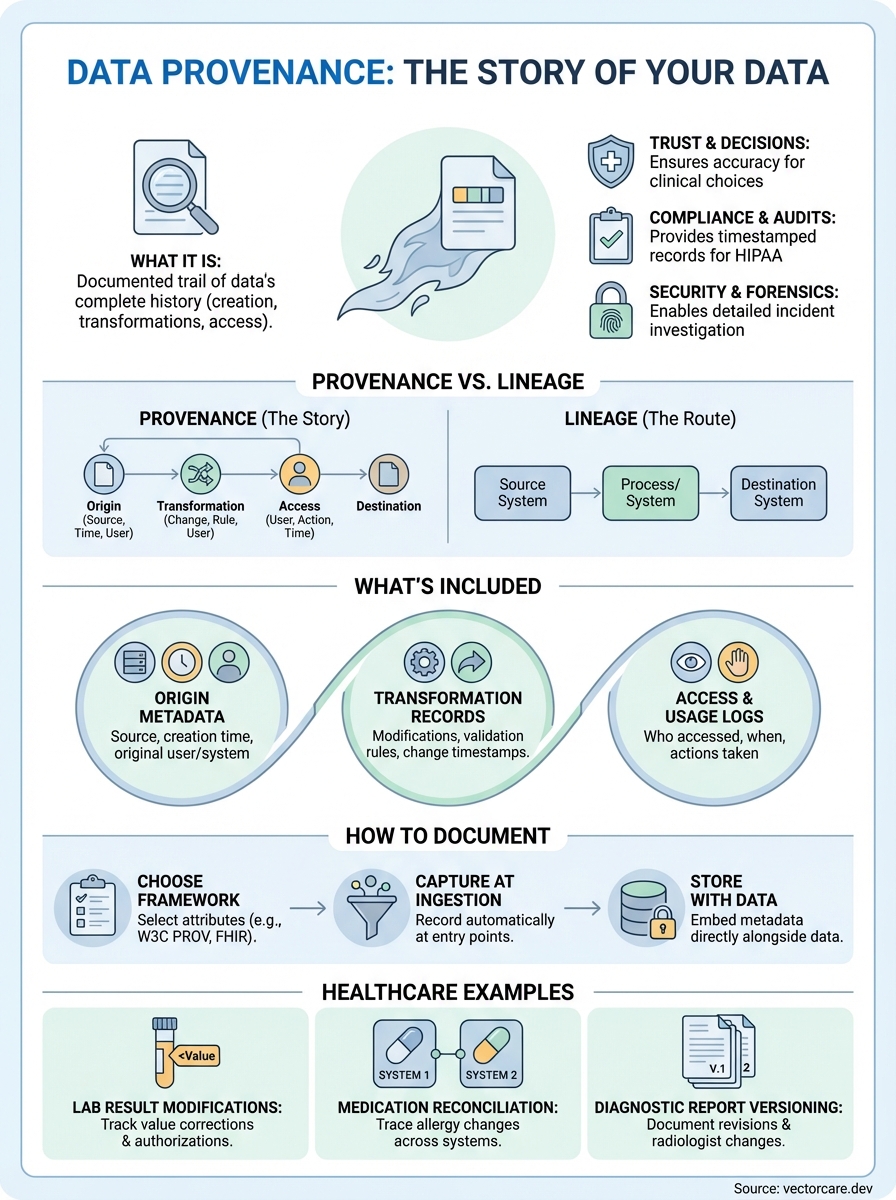
Every piece of clinical data in an electronic health record has a story, where it originated, who touched it, and how it changed along the way. What is data provenance? It's the documented trail that tells that story, capturing the complete history of data from its creation through every transformation it undergoes. For healthcare vendors building applications that integrate with systems like EPIC, understanding data provenance isn't optional, it's foundational to maintaining trust and compliance.
When patient information flows between systems, providers need confidence that the data they're acting on is accurate, unaltered, and traceable. This is precisely why platforms like VectorCare prioritize data integrity within SMART on FHIR workflows, because clinical decisions depend on knowing exactly where data came from and whether it can be trusted.
This article breaks down the definition of data provenance, explains how it differs from data lineage (a common point of confusion), and provides concrete examples of why it matters for data quality, security, and research validity. By the end, you'll have a clear framework for understanding how provenance tracking protects both your organization and the patients you serve.
Why data provenance matters
Understanding what is data provenance becomes critical the moment you realize that healthcare decisions carry life-or-death consequences. When a clinician orders medication based on lab results, prescribes treatment based on diagnostic imaging, or adjusts care plans based on patient-reported outcomes, they assume the data they're viewing is accurate and unmodified. Without provenance tracking, you have no way to verify that assumption or investigate when something goes wrong.
Clinical decision accuracy depends on data trust
Your application might pull allergy information from an EHR, display it to a provider, and use it to recommend treatments. If that allergy data was incorrectly transcribed during a system migration three years ago, and no provenance record exists to flag the discrepancy, you've just created a patient safety risk. Provenance tracking lets you trace the data back to its original source, identify where errors entered the pipeline, and determine whether the information you're displaying can be trusted for clinical use.
Data without documented provenance is data without accountability, and in healthcare, accountability isn't optional.
Compliance requires audit trails
HIPAA regulations demand that you maintain detailed records of who accessed patient data, when they accessed it, and what they did with it. Provenance documentation provides exactly that audit trail, creating a timestamped record of every interaction with sensitive information. During compliance audits or legal proceedings, you need to demonstrate that your application handles data responsibly. Provenance records give you concrete evidence of data handling practices, showing regulators that you've maintained appropriate controls throughout the data lifecycle.
Security incidents need forensic clarity
When a potential data breach occurs, your incident response team needs to answer specific questions quickly. Which records were accessed? Were they modified or simply viewed? How far did potentially compromised data spread through your systems? Strong provenance tracking gives you the forensic evidence needed to scope the incident accurately, notify affected parties appropriately, and implement targeted remediation. Without it, you're forced to assume worst-case scenarios and report breaches that may have been contained or never occurred.
Data provenance vs data lineage
People often confuse these terms because both relate to tracking data, but they serve fundamentally different purposes. Understanding the distinction helps you implement the right monitoring strategy for your healthcare application and avoid gaps in your data governance framework.

Data lineage tracks flow
Data lineage shows you the path data takes through your systems, mapping how information moves from source to destination. Think of it as a pipeline diagram that visualizes which systems, processes, and transformations your data passes through. When patient demographics flow from your intake form through your application and into EPIC, data lineage documents that journey at a high level. You see the route, but you don't necessarily see the detailed changes made along the way.
Data provenance documents history
Provenance goes deeper by capturing the complete record of what happened to the data at each step. While lineage tells you that lab results moved from your external vendor into the EHR, provenance records who entered the data, when they entered it, what validation rules were applied, whether values were modified, and who approved those modifications. When you ask what is data provenance, you're asking about the detailed audit trail that proves data integrity, not just the flow chart showing where data traveled.
Data lineage tells you the route; data provenance tells you the story of everything that happened along the way.
Your application needs both. Lineage helps you troubleshoot integration issues and understand system dependencies. Provenance gives you the evidence needed for compliance audits, error investigation, and maintaining trust in clinical data quality.
What data provenance includes
When you track data provenance in your healthcare application, you're documenting specific elements that together create a complete audit trail of information. These components work together to answer the fundamental questions about any piece of clinical data: where did it come from, who modified it, and can you trust it for clinical use? Understanding what is data provenance means knowing exactly which metadata elements you need to capture and maintain throughout the data lifecycle.

Origin metadata
Your provenance record starts by capturing the source of the data and the circumstances of its creation. This includes the originating system (was it entered directly into your application, pulled from an external lab system, or imported from a patient portal?), the timestamp of creation, and the user or process that generated it. You also document the format and version of the source system, which becomes critical when troubleshooting data quality issues that trace back to specific software versions or configuration changes.
Transformation records
Data rarely moves through your systems unchanged. Provenance tracking captures every modification made to the information, including the nature of the transformation (format conversion, value mapping, validation corrections), the logic or rules applied, and the timestamp of each change. When lab results undergo unit conversion or diagnostic codes get mapped between classification systems, you need records showing exactly what changed and why.
Complete transformation records turn your data pipeline from a black box into a transparent, auditable process.
Access and usage logs
Provenance documentation includes records of who accessed the data, when they viewed or modified it, and what actions they took. This creates the accountability trail required for HIPAA compliance and security investigations, showing which users, applications, or automated processes interacted with each piece of information throughout its lifecycle.
How to document data provenance
Documenting data provenance requires a systematic approach that captures metadata at every point where data enters, transforms, or moves through your application. Your strategy needs to balance comprehensive tracking with operational efficiency, ensuring you collect enough detail to support audits and investigations without creating storage or performance bottlenecks. The key is establishing clear standards for what gets captured and building those requirements directly into your data pipeline architecture from the start.
Choose your metadata framework
Your first step is selecting the specific attributes you'll track for every piece of data. Start with fundamental elements like source system, creation timestamp, and originating user, then add transformation details such as validation rules applied, format conversions performed, and modification timestamps. Healthcare applications often adopt standards like W3C PROV or HL7 FHIR Provenance resources, which provide structured frameworks for organizing this metadata consistently across different data types and systems.
Capture at ingestion points
The most critical moment for provenance tracking happens when data enters your system. Build capture mechanisms into every ingestion point, whether that's a user input form, an API endpoint receiving external data, or a batch import process. Your application should automatically record source details and initial context without requiring manual documentation. This ensures no data flows through your systems without a clear origin record, which becomes essential when you need to trace quality issues back to their source.
Automated provenance capture at ingestion points eliminates the gaps that manual documentation inevitably creates.
Store metadata with the data
Rather than maintaining separate provenance records, embed metadata directly alongside the data it describes. This approach keeps provenance information accessible whenever the data gets accessed, queried, or displayed, ensuring your audit trail moves with the information it documents.
Data provenance examples in healthcare
Seeing what is data provenance in action helps clarify why this concept matters for clinical applications. Real-world healthcare scenarios demonstrate how provenance tracking prevents errors, supports investigations, and maintains data integrity across complex workflows. These examples show the specific metadata you need to capture and how it protects both patients and your organization.
Lab result modifications
Your application receives a glucose test result from an external laboratory: 450 mg/dL. Before displaying this critical value to a provider, your system captures provenance metadata including the source lab (LabCorp), the performing location, the collection timestamp, and the analyzing technician's ID. Later, a medical assistant notices the decimal point should have been 45.0 mg/dL and corrects the error. Your provenance record documents who made the correction, when they made it, the original value, and the authorization workflow that approved the change. When the provider questions why the value differs from the printed lab report, you can show the complete audit trail proving the correction was legitimate and properly authorized.
Provenance records transform data corrections from suspicious changes into transparent, accountable improvements.
Medication reconciliation across systems
A patient's allergy list originates in your intake application, transfers to EPIC during admission, gets updated by the pharmacy team, and returns to your application for discharge planning. Provenance tracking captures each system that touched the data, every modification made, and which users authorized changes. During a medication error investigation, you trace the allergy data back through each transformation, discovering that a pharmacy technician accidentally removed a penicillin allergy during a system upgrade. Your detailed provenance trail identifies exactly when the error occurred and which patient records require correction.
Diagnostic report versioning
Radiology reports often undergo multiple revisions as radiologists review images more carefully or receive additional clinical context. Your provenance system documents each version of the report, the timestamp of each revision, the radiologist who made changes, and whether the report was marked as preliminary, amended, or final at each stage. This version history prevents providers from acting on outdated preliminary findings while ensuring they can access historical versions when tracking how diagnostic impressions evolved over time.
Key takeaways
Understanding what is data provenance gives you the foundation for building trustworthy healthcare applications that maintain complete audit trails from data creation through every transformation. Your provenance documentation captures source details, modification records, and access logs that together create the transparency required for clinical decision-making and regulatory compliance.
The difference between provenance and lineage matters practically. You need both to run secure, auditable systems, but provenance provides the detailed evidence that supports investigations, prevents errors, and proves your application handles patient data responsibly.
Building provenance tracking into your integration strategy from the start saves you from retrofitting audit capabilities later. When you're developing SMART on FHIR applications for EPIC, platforms like VectorCare handle the technical complexities of compliance and data integrity so you can focus on delivering clinical value rather than building infrastructure.
The Future of Patient Logistics
Exploring the future of all things related to patient logistics, technology and how AI is going to re-shape the way we deliver care.