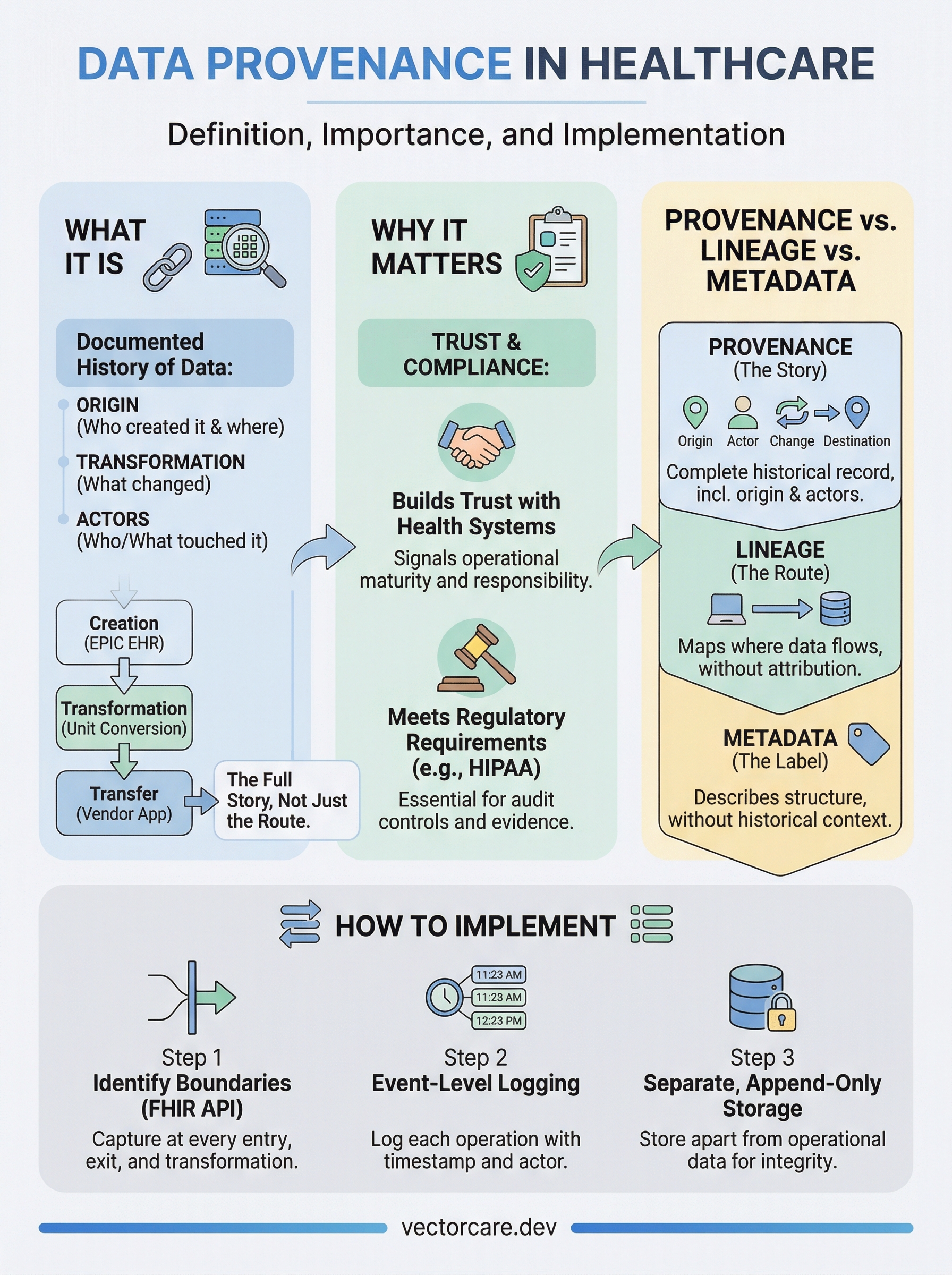
Data Provenance Definition: What It Is and Why It Matters


Every time a patient's record moves between systems, from an EPIC EHR to a vendor application and back, someone needs to answer a basic question: where did this data come from, and what happened to it along the way? That question sits at the heart of the data provenance definition, a concept that has become essential for anyone building or operating within healthcare IT. Data provenance is the documented history of a piece of data: its origin, every transformation it undergoes, and every system or person that touches it.
For healthcare vendors integrating with EHR platforms like EPIC, understanding data provenance isn't academic, it's operational. When your application pulls patient demographics, lab results, or clinical notes through FHIR APIs, you inherit a responsibility to track how that data was sourced and used. Auditors, compliance teams, and health systems all expect it. HIPAA demands it. And the more your product handles protected health information across clinical workflows, the more provenance matters.
This is exactly the kind of challenge VectorCare was built to address. Our no-code platform handles the technical complexity of EPIC integration, including SMART on FHIR compliance and HIPAA-ready architecture, so vendors can focus on their core product rather than data plumbing. But whether you build your integration with us or from scratch, you need to understand data provenance to do it right.
This article breaks down what data provenance means, why it matters for data integrity and regulatory compliance, how it differs from data lineage, and how it applies to healthcare data moving through EHR ecosystems.
What data provenance is and what it records
At its core, the data provenance definition describes the complete record of a dataset's life: where it was created, who created it, how it moved, and what changed along the way. Think of it like a chain of custody document used in legal proceedings. Every step in that chain is logged, timestamped, and attributed to a specific actor or system. For healthcare vendors, this means every patient record, clinical observation, or lab value your application reads or writes carries an implicit history that you are responsible for maintaining.
Provenance is not just a record of where data came from; it is a record of everything that shaped what the data became.
The origin layer: where data starts
Origin is the first thing provenance tracks. A provenance record captures the source system (such as an EPIC EHR instance), the timestamp of creation, the identity of the user or process that generated the data, and the context in which it was collected. In clinical settings, this might be a nurse entering vitals at the point of care, an automated device pushing telemetry, or a billing system importing a diagnosis code. Each of these origins carries a different level of reliability and a different set of audit requirements.
When you build an application that reads FHIR resources from EPIC, the origin metadata often travels with those resources. HL7's FHIR standard includes a dedicated Provenance resource specifically designed to capture this information, including the agent (who or what recorded the data), the entity (what was recorded), and the recorded timestamp. Ignoring this layer means losing critical context that auditors and clinical staff depend on to verify data accuracy.
Transformations, transfers, and timestamps
Data rarely stays in its original form. Every transformation, whether it is a unit conversion, a field mapping, a de-identification step, or an aggregation, is something provenance should record. Alongside transformations, provenance tracks transfers between systems: when your application sends patient data to a third-party analytics platform or receives updated records from a payer, those handoffs need to be logged with timestamps and the identities of the systems involved.

This level of detail is what saves you when something goes wrong. If a lab value arrives at your application with the wrong unit, provenance gives you a clear path back to the exact point where the error occurred. Without those records, diagnosing data quality issues becomes a slow, manual process that can take days and still leave your team without a definitive answer.
Why data provenance matters for trust and compliance
When a health system evaluates your application for contract consideration, they ask one fundamental question: can we trust this vendor with our patients' data? A strong data provenance definition and the practices that support it give you a concrete, auditable answer to that question. Without provenance records, you are asking health systems to take your word for how data moves through your system. With them, you hand over documented evidence that every record is tracked, attributed, and protected from the moment it enters your application.
Building trust with health systems
Health systems run their own security and privacy reviews before onboarding any vendor. Your ability to show clear provenance records during those reviews signals operational maturity. It tells clinical and IT leadership that your platform does not just process patient data. It accounts for that data at every step, which reduces the risk that the health system absorbs by working with you.
Provenance is one of the fastest ways to move from a vendor a health system is curious about to one they are confident in.
Meeting regulatory requirements
HIPAA requires covered entities and business associates to maintain audit controls that record and examine access to electronic protected health information. If your application touches FHIR resources from EPIC, you are almost certainly operating as a business associate, which means those audit requirements apply to you directly. Provenance records satisfy a significant portion of that obligation by logging who accessed what data, when, and why. Regulators reviewing a breach or complaint will look for exactly this kind of documentation, and gaps in that record are treated as violations regardless of whether a breach actually occurred.
Data provenance vs lineage vs metadata
People use data provenance, data lineage, and metadata interchangeably, but each term covers a distinct layer of information about data. Confusing them creates real gaps in your compliance documentation and makes diagnosing data quality problems far harder than it needs to be.

Data lineage: the path, not the story
Data lineage tracks where data flows across systems, from source to destination, mapping how datasets connect and how transformations link them. It answers "how did data get here?" at the pipeline level and helps you audit data dependencies across your infrastructure.
Lineage does not capture who created the data, under what authority, or why specific values changed at specific points in time. It shows movement without attributing responsibility.
Lineage shows you the route; provenance tells you the full story of the journey.
Metadata: description without history
Metadata describes data's structure and attributes, including field names, data types, and schema definitions. Systems use it to interpret and index information correctly. On its own, metadata carries no historical context: it tells you that a field holds an ISO 8601 date, not who entered that value, when, or under what clinical circumstances.
Your metadata strategy and your provenance strategy need to work together, not substitute for each other.
Where the data provenance definition fits
The data provenance definition sits above both lineage and metadata. It incorporates origin attribution, transformation history, and actor identity into one connected record that the other two concepts cannot provide on their own.
Think of lineage as the map of a pipeline, metadata as the label on a container, and provenance as the complete account of every event that shaped what ended up inside it. A sound data governance framework needs all three working in parallel.
Types, standards, and granularity
Not all provenance records look the same. The data provenance definition you apply in practice depends on what type of provenance you capture, which standards your stack must comply with, and how granular your records need to be to satisfy both operational and regulatory requirements.
Prospective vs. retrospective provenance
Prospective provenance captures a plan or intent before data is generated, recording the expected workflow, parameters, and agents involved. Retrospective provenance records what actually happened after the fact, logging the real origin, transformations, and actors that touched the data. For healthcare vendors, retrospective provenance is the baseline requirement because regulators and auditors want evidence of what occurred, not just what was planned.
In most healthcare compliance audits, retrospective provenance records are the only kind that count as evidence.
Standards that govern provenance
Two standards shape how most teams implement provenance in healthcare data systems. The W3C PROV specification defines a general-purpose data model for representing provenance across any domain, using entities, activities, and agents as its core building blocks. HL7's FHIR Provenance resource applies that same conceptual structure specifically to clinical data, letting you attach provenance records directly to FHIR resources like Patient, Observation, and DiagnosticReport. If your application integrates with EPIC, the FHIR Provenance resource is the format your provenance records should follow.
Granularity: how detailed your records need to be
Coarse-grained provenance logs data at the dataset or document level, which is fast and low-overhead but may miss critical detail. Fine-grained provenance tracks individual fields and values, which gives you the precision to pinpoint exactly where a specific value changed. For high-risk clinical data like medication orders or diagnostic results, fine-grained records are worth the added storage cost.
How to implement data provenance in modern stacks
Applying the data provenance definition in a real stack means making deliberate choices about where you capture records, what format you use, and how you connect provenance data to your operational logs. Start by identifying every system boundary in your data flow: each point where data enters, exits, or transforms is a location where a provenance event should fire. For healthcare vendors working with EPIC, those boundaries include the FHIR API call, any internal transformation layer, and every outbound integration to a third-party service.
The most common implementation gap is capturing provenance at ingestion but skipping it at transformation, leaving your audit trail incomplete exactly where errors tend to occur.
Log at the event level, not the batch level
Event-level logging records provenance as each individual operation happens rather than aggregating records after the fact. This approach gives you a complete, timestamped trail that maps directly to the FHIR Provenance resource structure and satisfies HIPAA audit control requirements without manual reconstruction.
When you log at the batch level, you lose the granularity auditors need to verify specific field changes during a compliance review. Open frameworks like Apache Atlas provide event-level provenance capture across data pipelines without requiring you to build custom instrumentation from scratch.
Store provenance separately from operational data
Your provenance records should live in a dedicated store that your application's normal read and write operations cannot overwrite. Use append-only storage so records accumulate without the risk of accidental deletion, and apply access controls that restrict provenance log queries to authorized roles only. Keeping provenance isolated also makes retrieval straightforward during a compliance audit, when both speed and accuracy matter.
Bring provenance into daily work
Treating the data provenance definition as a one-time compliance checkbox misses the point. Provenance earns its value when your team builds it into every integration decision, from the first FHIR API call to the final record write. Start with your highest-risk data flows, clinical observations, medication records, and diagnostic results, then expand from there. Document your origin sources, log transformations at the event level, and store provenance records in an append-only store that auditors can access without disrupting your live system.
For healthcare vendors building on EPIC, getting provenance right from the start saves significant remediation work later. VectorCare's no-code platform handles SMART on FHIR compliance and HIPAA-ready architecture out of the box, so you spend time on your core product instead of data plumbing. If you are ready to build an EPIC integration with compliance built in from day one, see how VectorCare accelerates your EPIC integration.
The Future of Patient Logistics
Exploring the future of all things related to patient logistics, technology and how AI is going to re-shape the way we deliver care.