What Is USCDI? A Practical Guide to Health Data Interop


What Is USCDI? A Practical Guide to Health Data Interop
If you're a healthcare vendor looking to integrate with EPIC or other EHRs, you've likely encountered the term USCDI. But what is USCDI, exactly, and why should it matter to your product strategy?
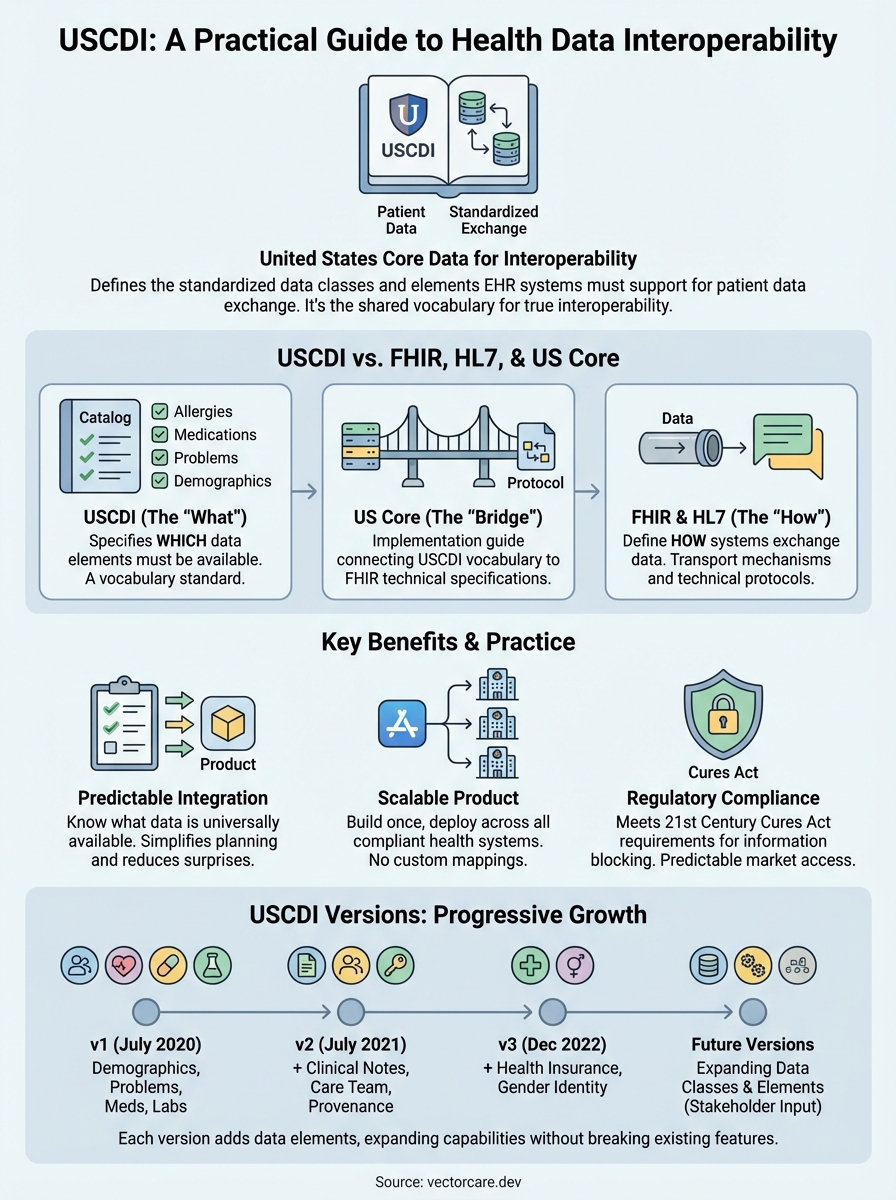
The United States Core Data for Interoperability defines the standardized health data classes and elements that EHR systems must support for patient data exchange. Unlike FHIR or HL7, which are technical protocols for how data moves, USCDI specifies what data needs to be exchangeable. It's the shared vocabulary that makes true healthcare interoperability possible.
For vendors building applications that embed into clinical workflows, like the SMART on FHIR apps we help deploy at VectorCare, USCDI compliance determines which patient data your solution can reliably access across health systems. Understanding these data requirements upfront saves development time and prevents costly integration surprises.
This guide covers USCDI's purpose, key differences between versions, the specific data elements it includes, and how it works alongside FHIR to power modern EHR integrations.
Why USCDI matters for interoperability
Before USCDI existed, each EHR vendor decided which patient data fields to expose and how to label them. Your integration team might discover that one health system's "Problem List" mapped to another's "Condition List," or that medication data structures varied wildly between implementations. This inconsistency forced vendors to build custom mappings for every health system, ballooning both development time and ongoing maintenance costs.
USCDI solves the fragmentation problem
The Office of the National Coordinator for Health Information Technology created USCDI to establish a common baseline for what data elements every certified EHR must support. When you build an integration against USCDI standards, you know that all certified systems will provide the same core patient information in predictable formats. This standardization eliminates the guesswork from your technical planning.
Health systems also benefit because they no longer need to customize their EHR for every vendor integration. They configure their system to support USCDI once, and any compliant application can then access that data. This mutual understanding of what is USCDI accelerates procurement cycles and reduces implementation friction.
USCDI transforms healthcare integration from a custom engineering project for each health system into a scalable product deployment.
Your integration becomes predictable
Understanding USCDI requirements during your product design phase prevents costly surprises later. If your application relies on specific patient data like care team information or provenance details, you can verify whether USCDI includes those elements before committing engineering resources. Version planning becomes straightforward because you know which data classes will be available across your target health systems.
USCDI also protects your long-term product roadmap. As new versions add data elements, you can anticipate which enhanced features will become universally accessible. This visibility helps you prioritize development work that aligns with industry-wide data availability rather than one-off health system capabilities.
Regulatory compliance built in
The 21st Century Cures Act mandates that certified EHR systems support USCDI for information blocking regulations. This means health systems must make USCDI data elements available to patients and authorized applications without special effort. Your application can request this data through standard APIs, and health systems face regulatory pressure to provide it.
For vendors, this regulatory framework translates into predictable market access. You don't need to negotiate separate data access agreements or navigate proprietary APIs for basic patient information. USCDI compliance becomes part of the health system's certification requirements, creating a level playing field where your technical capabilities matter more than your vendor relationships.
USCDI basics: data classes and data elements
When you explore what is USCDI in practice, you'll find it organizes health information into two structural layers: data classes and data elements. This hierarchy helps your development team understand both the broad categories of patient information available and the specific fields within each category. Data classes represent major groupings like medications or allergies, while data elements pinpoint the exact information fields your application can request.

Data classes organize patient information
USCDI defines data classes as the top-level categories that structure patient records. Your integration accesses information through classes like Patient Demographics, Problems, Medications, Procedures, and Laboratory Results. Each class represents a distinct clinical domain that health systems maintain in their EHR systems.
Think of data classes as the chapters in a patient's medical record. When your application needs allergy information, you query the Allergies and Intolerances data class rather than searching through unstructured notes. This categorical approach makes your API requests more efficient because you target specific information types rather than pulling entire patient records.
Data elements define the specific fields
Within each data class, data elements specify the individual data points you can retrieve. The Patient Demographics class, for example, includes data elements like name, birth date, sex, and preferred language. Your application receives these standardized fields regardless of which health system's EHR you're querying.
Data elements transform abstract categories into concrete API responses your code can process.
Consider the Medications data class. Its data elements include medication name, dosage, route, and frequency. When you build features that rely on medication data, you know these specific fields will be available across all USCDI-compliant systems. This predictability eliminates the conditional logic needed to handle system-specific variations in data availability.
The structure matters because your integration code maps directly to these elements. You write functions that expect specific data element responses, and USCDI ensures those responses arrive in consistent formats.
USCDI versions and what changes over time
Understanding what is USCDI includes recognizing that it evolves through numbered versions rather than remaining static. The Office of the National Coordinator releases new versions periodically to expand the baseline data requirements that certified EHR systems must support. Each version adds data classes and elements based on stakeholder input from healthcare providers, vendors, and standards organizations.
Your integration strategy needs to account for version timing because health systems adopt new USCDI versions according to EHR certification cycles. When ONC publishes a new version, it doesn't immediately appear across all health systems. The regulatory timeline typically gives EHR vendors time to implement changes before health systems must comply.
Version progression and new data elements
USCDI v1, published in July 2020, established the initial baseline with essential data classes like Patient Demographics, Problems, Medications, and Laboratory Results. Version 2, released in July 2021, added Clinical Notes, Care Team Members, and Provenance information. These additions responded to real-world integration needs that vendors and health systems identified during v1 implementation.
Version 3, published in December 2022, expanded coverage with Health Insurance Information, Laboratory Result Observation Status, and Patient Gender Identity. Each new version builds on previous requirements rather than replacing them. Your application that works with v2 data elements continues functioning when health systems upgrade to v3, while gaining access to the newly added fields.
USCDI versions expand your integration capabilities without breaking existing functionality.
The United States Core Data for Interoperability Task Force (USCDI+ TF) reviews submissions for future data elements through a public comment process. You can monitor proposed additions to plan your product roadmap around data that will become universally available. This transparency helps your team prioritize features that align with upcoming interoperability standards rather than custom one-off requests.
USCDI vs FHIR, HL7, and US Core
When you first encounter what is USCDI alongside terms like FHIR, HL7, and US Core, they can seem interchangeable. However, these standards serve fundamentally different purposes in your integration architecture. Understanding where each fits prevents technical confusion during your development planning and helps you choose the right tools for specific integration challenges.

The "what" versus "how" distinction
USCDI specifies which data elements your application should be able to access, like patient allergies or medication lists. It's a vocabulary standard that answers "what information exists in a patient record?" Your team references USCDI when planning features that depend on specific patient data fields.
FHIR and HL7, by contrast, define how systems exchange that data. FHIR provides the technical protocol for structuring API requests and responses, while HL7 v2 handles message-based transactions. These are transport mechanisms that move information between systems. Your developers work with FHIR resources to actually retrieve the data elements that USCDI defines.
USCDI tells you what patient data must be available, while FHIR and HL7 tell you how to retrieve it.
Where US Core bridges standards
US Core creates the practical connection between USCDI's vocabulary and FHIR's technical specifications. It's an implementation guide that maps each USCDI data element to specific FHIR resources and profiles. When your integration needs to access USCDI-required data through FHIR APIs, US Core provides the exact resource definitions and search parameters to use.
Your development team relies on US Core profiles to structure API calls that retrieve USCDI data elements. For example, US Core's Patient profile defines how to request the name, birth date, and other demographic elements that USCDI requires. This mapping removes ambiguity about which FHIR resources correspond to USCDI requirements.
How teams apply USCDI in practice
Your development workflow changes when you understand what is USCDI requires from your integration. Rather than building custom data mappings for each health system, you design your application around USCDI data classes from the start. This approach transforms your technical architecture from reactive problem-solving into proactive standards compliance.
Designing API calls around USCDI requirements
Your engineering team starts by mapping your application's data needs to specific USCDI elements. If your workflow requires patient medication information, you reference the Medications data class to identify which standardized fields you can request. This planning phase determines your API structure before you write any integration code.
When building SMART on FHIR apps, you structure your scope requests based on USCDI coverage. Your authorization requests specify the FHIR resources that correspond to required USCDI elements, like patient/MedicationRequest.read for medication data. This alignment ensures your application requests only the standardized data that health systems must provide.
Your integration code becomes portable across health systems when it requests USCDI-defined data rather than vendor-specific fields.
Validating data element coverage
Testing your integration involves verifying that you correctly retrieve all required USCDI elements from the EHR's FHIR API. Your quality assurance process checks whether responses include expected data element structures like medication dosage, frequency, and route. Missing elements indicate either incomplete API implementation or incorrect resource queries.
Your team also validates data format consistency across different health system environments. USCDI compliance means a patient's allergy information should arrive in the same structure whether you're integrating with EPIC, Cerner, or another certified EHR. This consistency testing confirms that your application logic handles USCDI data uniformly regardless of the underlying system.
Next steps
Understanding what is USCDI gives you the foundation for planning compliant EHR integrations that work across health systems. Your next move depends on where you are in your integration journey: researching requirements, designing your data model, or actively building SMART on FHIR applications.
Start by mapping your application's data requirements to specific USCDI data classes and elements. This exercise reveals which patient information your solution can reliably access and helps you identify any gaps between your planned features and available standardized data. Document these mappings early to guide your technical architecture.
If you're ready to build your integration without managing the technical complexity yourself, VectorCare's no-code platform handles USCDI compliance, FHIR implementation, and EPIC certification automatically. Your team configures workflows through a visual interface while we manage the standards compliance, hosting, and ongoing maintenance that keep your application running smoothly across health systems.
The Future of Patient Logistics
Exploring the future of all things related to patient logistics, technology and how AI is going to re-shape the way we deliver care.