Consent Management Definition: Process, Tools & Compliance


Every time a healthcare application pulls patient data from an EHR, there's a critical question behind the scenes: did the patient agree to this? That question sits at the core of the consent management definition, the structured process of collecting, storing, and honoring a person's preferences about how their data is used. For healthcare vendors building integrations with systems like EPIC, getting consent management right isn't optional. It's a compliance requirement and a trust issue.
Consent management covers more than just a checkbox on a form. It involves specific tools, workflows, and policies that govern data access across every touchpoint in a patient's care journey. Regulations like HIPAA, GDPR, and state-level privacy laws each impose their own rules, and the penalties for mishandling consent are steep, both financially and reputationally. For vendors working to embed their solutions into clinical workflows through EHR integrations, understanding these mechanics is foundational.
At VectorCare, we build no-code SMART on FHIR applications that connect healthcare vendors to EPIC EHR systems. Consent workflows are baked into that process, from HIPAA-compliant data exchange to configurable decision logic that respects patient preferences at every step. This article breaks down what consent management actually means, how the process works in practice, the tools that support it, and what compliance looks like when you're operating inside a health system's ecosystem.
Consent management definition and key terms
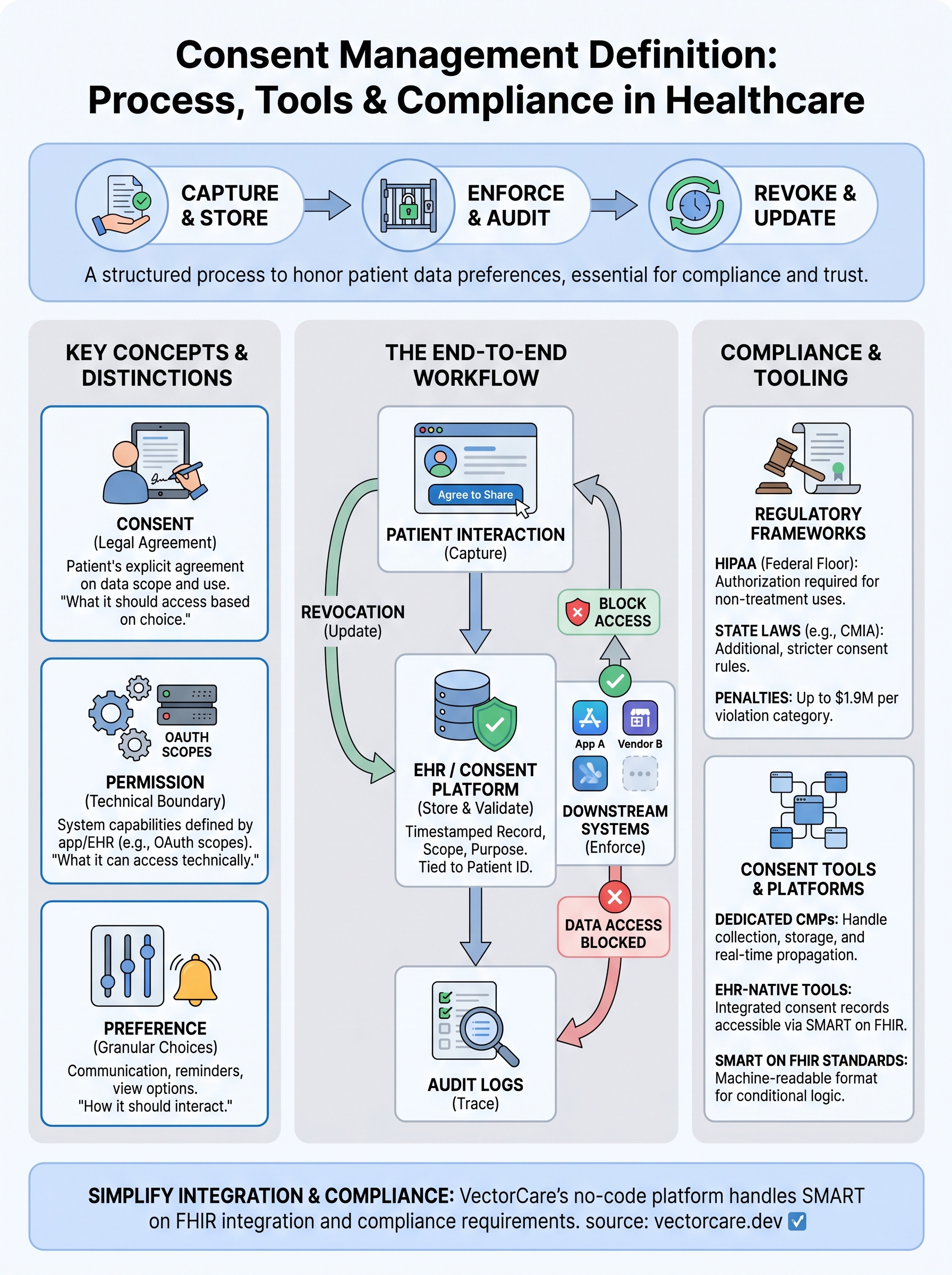
The consent management definition at its most basic level is: a system that captures a person's explicit agreement to share or use their data, then enforces that decision everywhere the data flows. In healthcare, this gets more specific. Patient consent determines which providers, vendors, and applications can access protected health information (PHI), and under what conditions. The system doesn't just record a "yes" or "no." It tracks the scope, timing, and revocability of that agreement.
What consent management covers
Consent management operates across three layers: data collection, enforcement, and auditing. When a patient agrees to share their records with a vendor's application, that agreement has to be logged with a timestamp, tied to a specific data scope, and stored in a way that's retrievable for audits. If the patient later revokes that consent, every downstream system receiving that data needs to reflect the change. In EPIC integrations, this means your application has to read and respect consent flags set within the EHR before pulling any patient data.
Your consent system also has to handle edge cases that show up in real clinical environments, such as minors, guardians, and emergency access scenarios. These situations require conditional logic in your workflow, not just a binary yes-or-no field. SMART on FHIR standards provide the technical framework for expressing these nuances in a machine-readable format that EHR systems like EPIC can interpret and enforce.
Consent is not a one-time event. It's an ongoing record that must stay in sync across every system that touches patient data.
Key terms in consent management
Understanding the core vocabulary helps you build compliant workflows from the start. Below are the terms that come up most often when working inside healthcare data environments:
| Term | What it means |
|---|---|
| Data subject | The individual whose data is being collected and used |
| Consent record | A time-stamped log of when and what a person agreed to |
| Scope of consent | The specific data types and uses covered by the agreement |
| Revocation | A person's right to withdraw consent at any time |
| Authorization | A broader permission granted under HIPAA for specific disclosures |
| Purpose limitation | The rule that data can only be used for the purpose stated at collection |
Each of these terms appears in regulatory frameworks like HIPAA and GDPR, and in the technical specs for SMART on FHIR applications. Knowing how they connect helps you design applications that don't just pass a compliance check on day one, but stay compliant as patient preferences change over time.
Why consent management matters for compliance
Compliance is the most direct reason healthcare vendors treat consent management seriously. Under HIPAA, any application that accesses, transmits, or stores protected health information (PHI) must operate within a strict framework of patient rights, including the right to control how their data is used. If your application touches patient records inside an EPIC environment, violations carry real financial consequences: civil penalties up to $1.9 million per violation category per year, and criminal liability in cases involving willful neglect.
HIPAA and the healthcare consent baseline
HIPAA sets the federal floor for consent in healthcare. It distinguishes between treatment-related disclosures, which generally don't require explicit patient consent, and uses of PHI for third-party vendor access or research, which do. For vendors building SMART on FHIR integrations, the authorization rules under HIPAA's Privacy Rule govern exactly what data your application can request and under what conditions. A valid HIPAA authorization must include all of the following:
- The specific data types being disclosed
- The stated purpose of the disclosure
- An expiration date or triggering condition
- The patient's signature and the date signed
Getting the consent management definition right inside your application architecture prevents HIPAA violations before they happen, rather than correcting them after a compliance audit.
State-level privacy laws expand your obligations
Federal law is not the only framework you need to account for. State privacy laws like California's Confidentiality of Medical Information Act (CMIA) layer additional consent requirements on top of HIPAA's baseline. Some states require explicit opt-in consent for data categories that HIPAA treats as routine treatment disclosures. If your vendor platform serves health systems across multiple states, your consent workflows need to handle those jurisdictional variations without requiring manual reconfiguration at each new deployment. Building that flexibility into your consent logic from the start saves significant rework down the line.
How consent management works end to end
The consent management definition describes a process, but understanding that process step by step shows you where the real complexity lives. Consent doesn't flow through a single system. It moves across your application, the EHR, your backend, and any downstream vendor receiving patient data. Each handoff is a point where consent records must be validated and enforced before any data transfer happens. Skipping that validation at even one step creates a compliance gap that auditors will find.

Capturing and storing consent
When a patient interacts with your application, the first step is consent capture: presenting the patient with a clear description of what data your application will access, why, and for how long. That agreement gets stored as a structured record tied to the patient's identity, a specific data scope, and a timestamp. In SMART on FHIR environments, this record often lives inside the EHR and gets referenced by your application at each data request.
Your application needs to request only the FHIR resource scopes that match what the patient agreed to. Requesting broader access than what was consented to is a compliance violation, regardless of whether you actually use the extra data.
Consent capture is only useful if your data request layer checks it before pulling records, not after.
Updating and revoking consent
Patients can change or revoke their consent at any time, and your system has to propagate that update immediately. A revocation logged in the EHR needs to block your application from making further data requests. Build your integration so it checks consent status at each session, not just once during onboarding. Logging every consent check with a timestamp gives you a defensible audit trail if a health system or regulator asks how your application handled a specific patient's preferences.
Consent, permission, and preference management
These three terms get used interchangeably in healthcare vendor conversations, but they describe distinct layers of control that your application needs to handle separately. Understanding where each one fits prevents you from building a system that handles one layer correctly while leaving the others exposed. The consent management definition applies specifically to the legal agreement a patient makes about their data, while permission and preference management handle different but related functions.
Permission management vs. consent management
Permission management controls what your application is technically allowed to do within a system, most often defined through OAuth scopes in a SMART on FHIR environment. When your app authenticates with EPIC, the scopes granted determine which FHIR resources your application can read or write. This is a technical boundary set by the health system or the EHR platform, not by the patient. Consent, by contrast, is a patient-level agreement that sits on top of those technical permissions. Your app might have permission to read medication records, but if a specific patient hasn't consented to share that data with your vendor, you should not pull it regardless of the scope your app holds.

Permission defines what your application can access. Consent defines what it should access based on patient agreement.
Preference management
Preference management handles the ongoing, granular choices patients make about their care experience, such as communication channels, appointment reminders, or which providers can view certain record types. These preferences often live inside the EHR as patient-recorded data and feed into your application's decision logic and routing rules. Unlike consent, preferences don't carry the same legal weight, but ignoring them damages patient trust and can conflict with consent conditions tied to specific use cases. Build your workflows so preference data updates trigger re-evaluation of downstream logic automatically, rather than requiring manual intervention each time a patient changes a setting.
Consent tools and platforms you can use
The right tooling makes the difference between a consent workflow that holds up under audit and one that creates gaps at every data handoff. When you're building healthcare vendor applications, the consent management definition you implement needs to be backed by platforms that can handle real-time enforcement, structured record storage, and cross-system propagation without manual intervention.
Consent management platforms for healthcare
Consent management platforms (CMPs) handle the collection, storage, and enforcement of patient consent records as a dedicated function. In healthcare contexts, these platforms need to support HIPAA-compliant data handling, granular scope tracking, and revocation propagation to connected systems. Some platforms also provide pre-built templates for HIPAA authorizations that satisfy the required elements without requiring your engineering team to build them from scratch. You can review the regulatory baseline for these requirements directly through the HHS HIPAA Privacy Rule guidance, which defines what a valid authorization must include.
A standalone CMP adds a defensible audit layer that your EHR integration alone cannot provide.
EHR-native consent tools and SMART on FHIR
EPIC's native consent functionality stores patient authorization records directly inside the EHR, making them available to SMART on FHIR applications at the point of data request. When your application authenticates with EPIC, it can query the patient's existing consent records before requesting any FHIR resources. This keeps your consent logic tightly coupled to the source of truth rather than maintaining a separate database that can fall out of sync.
Your application should log every consent check it performs, including the timestamp, patient ID, and data scope evaluated. Those logs are what you produce when a health system asks how your platform handled a specific patient's preferences during a compliance review.
Where to go from here
The consent management definition gives you the foundation, but applying it inside a real healthcare vendor workflow requires more than policy knowledge. You need a technical architecture that captures consent, enforces it at every data request, propagates revocations in real time, and produces a clean audit trail that satisfies both HIPAA requirements and health system security reviews. That architecture gets complicated fast when you're building from scratch, especially inside a SMART on FHIR environment where EHR integration adds layers of OAuth scopes, FHIR resource logic, and compliance checks on top of your core product.
Vendors who need to move faster and avoid rebuilding that infrastructure themselves have a direct path forward. VectorCare's no-code platform handles the SMART on FHIR integration layer, compliance requirements, and EPIC App Orchard submission so you can focus on your product. If you're ready to get your application into clinical workflows without the engineering overhead, explore what VectorCare builds for healthcare vendors.
The Future of Patient Logistics
Exploring the future of all things related to patient logistics, technology and how AI is going to re-shape the way we deliver care.